Abstract
Homologous recombination of mice during meiosis is critical for gamete formation. Recombination is initiated by programmed DNA double-strand breaks that occur preferentially at hotspots scattered throughout the genome. These double-strand breaks are repaired from the homologue, resulting in a crossover or uncrossover product. Multiple noncrossover events are required for homologous pairing, and at least one crossover is critical for proper chromosome segregation in the first meiotic division.
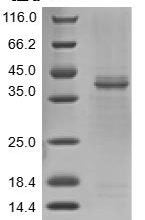
Consequently, homologous recombination in meiosis occurs at high frequencies. This chapter describes how to characterize crossing and non-crossing at a hotspot in mice by allele-specific PCR. Amplifying recombinant products directly from sperm DNA is a powerful approach for determining recombination frequencies and mapping recombination breakpoints, providing insights into homologous recombination mechanisms.
Exploring the double-strand break landscape in the mouse
DSBs are more likely to occur in some genomic regions than others. Elucidating the mechanisms that shape the DSB “landscape” requires identifying the biochemical factors involved, understanding how they influence Spo11, and breaking down their interactions. We developed a powerful method to address these issues: each Spo11 oligo is a tag that accurately records where a break occurred, so deep sequencing of the oligos allowed us to quantitatively map DSBs in the yeast genome with high resolution. of nucleotides and high sensitivity.
More recently, we extended this methodology to mice (Lange et al. 2016; Yamada et al. 2017). An important hotspot determinant in most mammals is PRDM9, which features a rapidly evolving histone methyltransferase module and Zn-finger DNA-binding domain. DNA-binding specificity defines breakpoints, but how PRDM9 targets SPO11 activity is unknown.
Furthermore, although the mammalian DSB landscape has a hierarchical shape at different size scales, as in yeast, studies tend to focus almost exclusively on hotspots, so we know little about other features. The much higher resolution and sensitivity of SPO11-oligo mapping compared to previous ssDNA sequencing (SSDS) maps allow us to address many of these challenges in ways that were not previously impossible.
Role of ATM in feedback control of double-strand break formation
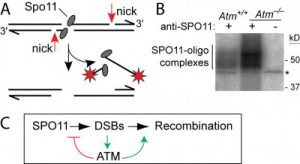
ATM is a mutated Ser/Thr kinase in ataxia-telangiectasia (A-T), a cancer-prone disease. DSB-activated ATM activates cell cycle checkpoints and promotes DNA repair in somatic cells, but ATM is also essential during normal, undisturbed meiosis for reasons that are unclear. A major breakthrough came from our discovery that Atm–/– spermatocytes experience very high numbers of DSBs.
Furthermore, the absence of ATM makes DSB formation extremely sensitive to the level of SPO11 expression, which likely explains why Spo11 heterozygosity partially rescues Atm deficiency: many defects in Atm-depleted spermatocytes are likely to be caused. by very high DSBs, which are decreased by reduced expression of SPO11. We proposed that ATM activation governs a negative feedback loop that restricts SPO11 to limit the number of DSBs (Lange et al., 2011). Independent studies in Drosophila (McKim lab) and yeast (Kleckner, Neale, Cha and Fung labs) led to a similar conclusion. More recently, we have explored how ATM shapes the DSB landscape in mice (Lange et al. 2016) and yeast (Mohibullah and Keeney 2017).
XY recombination
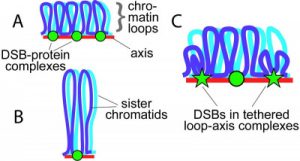
Sex chromosome segregation poses challenges for the male meiotic cell because X and Y share only a small region of homology, the pseudoautosomal region (PAR): at least one DSB must form within the PAR; that DSB must locate and hire its counterpart partner; and a junction should be formed. To understand how cells cope with these challenges, we studied the behaviour and structure of PAR in normal meiosis and explored the genetic requirements to ensure PAR DSB formation (Kauppi et al. 2011).
We found that mouse PAR DNA occupies unusually long chromosome axes, potentially as shorter chromatin loops, predicted to promote DSB formation. We also find that most PARs have delayed the appearance of RAD51 foci marking DSB ends, and provide evidence that PAR DSBs are genetically distinct from “global” DSBs. These findings uncover specific mechanisms that overcome the unique challenges of X-Y recombination.
Many questions remain, including What aspects of PAR structure and behaviour are intrinsic to PAR itself? Are pro-DSB factors specifically enriched in the PAR in men? One approach we are using to address these issues is to query by immuno-FISH the higher-order structure and protein composition of PAR at both male and female meiosis. In females, the X chromosomes synapse and recombine along their length as autosomes, and the PAR has a much lower crossing over rate than in males. Quantification of PAR RAD51 foci in oocytes will allow us to determine whether less frequent crossover reflects less DSB, a lower probability of a DSB becoming a crossover, or both.
Measuring the sizes of the spindles and loops will test the correlation between chromosome structure and DSB potential. Immuno-FISH will also allow us to test the hypothesis that PAR preferentially accumulates DSB-promoting factors. These and other studies of PAR compared to autosomes and in spermatocytes compared to oocytes will provide critical information about this important region of the genome and help us understand the mechanisms that meiotic cells exploit to promote precise chromosome segregation sexual.
Non-allelic homologous recombination
Recombination usually occurs between allelic sequences in homologues or between sister chromatids, but non-allelic homologous recombination (NAHR) between repetitive sequences can cause chromosomal rearrangements. In humans, germline NAHR represents more than 30 types of disease-causing mutations, with phenotypes including mental retardation and male infertility. Little is known about this process: What is the mechanism of meiotic NAHR and how does it differ from allelic recombination?
What strategies do cells employ to limit NAHR? From mouse SPO11-oligo maps, we discovered several repeats that contained strong DSB hotspots (Yamada et al. 2017; S. Kim and J. Lange, unpublished data). We are using PCR-based physical assays on sperm DNA to explore the mechanism and regulation of NAHR, comparing allelic and non-allelic recombination frequencies and testing the hypothesis that mismatch repair machinery inhibits NAHR. These studies will provide new insights into how cells minimize the risk of germline genome instability posed by the need to distribute recombination events in a genome that is riddled with repetitive elements.